Author: Richard G Ohye, MD, Head, Division of Pediatric Cardiovascular Surgery; Program Director, Pediatric Cardiac Surgery Fellowship, University of Michigan Medical Center
Coauthor(s): Ralph S Mosca, MD, Director, Pediatric Cardiac Surgery, Associate Professor, Department of Surgery, New York Presbyterian Medical Center; Edward L Bove, MD, Associate Director, PICU, CS Mott Children’s Hospital; Director, Department of Surgery, Section of Thoracic Surgery, Division of Pediatric Cardiovascular Surgery, Professor, University of Michigan Medical Center
Treatment of Hypoplastic Left Heart Syndrome (HLHS)
Medical Therapy
Initial medical support in infants with hypoplastic left heart syndrome (HLHS) requires a specific medical regimen. The goals of preoperative management are to maintain ductal patency and to provide the appropriate balance between the systemic and pulmonary vascular resistances. Intravenous prostaglandin E is infused at 0.05 mcg/kg/min to maintain patency of the ductus arteriosus. This dose may be titrated to keep the ductus arteriosus open while minimizing the risk of apnea.Oxygen saturation is monitored by pulse oximetry. Acidosis is rapidly reversed using sodium bicarbonate. The fraction of inspired oxygen (FIO2) is adjusted to maintain a relative hypoxemia (oxygen saturation 75-80%), which aids in preventing the pulmonary vasodilatation associated with high oxygen concentrations. Even in the neonate who is being resuscitated because of circulatory collapse, ventilation with a high concentration of oxygen is avoided because it may only further decrease pulmonary vascular resistance and systemic blood flow.
Blood transfusion should be performed to maintain the hematocrit between 45-50%. Mechanical ventilation is avoided when possible, but infants on ventilation may require sedation with intravenous fentanyl to prevent tachypnea. In patients with significant pulmonary overcirculation, hypoventilation to maintain a mild respiratory acidosis (partial pressure of carbon dioxide [PCO2] of 45-55 mm Hg) and elevation of pulmonary vascular resistance may be used. Occasionally, inhaled nitrogen or carbon dioxide can be added to reduce the FIO2 to between 16-18% to increase pulmonary vascular resistance. Inotropic support is advantageous in patients with depressed right ventricular function.
Nourishment is usually provided via intravenous hyperalimentation, which avoids the added risk of necrotizing enterocolitis prior to surgery. Diuretics are added, as necessary, when pulmonary congestion becomes apparent. This regimen simulates the fetal balance of pulmonary and systemic vascular resistance, stabilizing the infant while deciding on therapeutic options.
Surgical Therapy
Parents of children with HLHS are presented with the following 3 options: (1) supportive therapy only (leading usually to rapid demise), (2) staged reconstruction, and (3) orthotopic cardiac transplantation. Each institution must assess its results with the various modes of therapy and counsel the parents accordingly. As outcomes of palliative procedures and heart transplantation in patients with HLHS have improved, even surpassing therapies for other complex forms of congenital heart disease in some patients, the first option of supportive therapy only has been challenged. The techniques and results of staged reconstruction are discussed below.
The goal of staged reconstruction is a Fontan procedure, creating separate pulmonary and systemic circulations supported by a single (right) ventricle. The initial stage must provide unobstructed systemic blood flow from the right ventricle to the aorta and coronary arteries, relieve any obstruction to pulmonary venous return, and limit pulmonary blood flow by virtue of an appropriately sized systemic–to–pulmonary artery shunt or RVPAC.
As a result of the relatively high pulmonary vascular resistance present in the newborn period, a systemic shunt is necessary, and the right ventricle performs the increased volume of work of both the pulmonary and systemic circulations. Preservation of right ventricular function has been aided by using smaller initial aortopulmonary shunts to limit right ventricular volume overload, by using an RVPAC, and by using an interim procedure between the Norwood and Fontan operations. This staging procedure, either a bidirectional Glenn anastomosis or a hemi-Fontan procedure, is usually performed at age 6 months.
Recently, the use of an RVPAC as an alternative to the traditional MBTS has been proposed. Initially described by Norwood in his original description of the Norwood procedure, the use of the MBTS quickly became the preferred source of pulmonary blood flow. In 2002, Sano and colleagues described their experience with the Norwood procedure, with improvements in hospital survival from 53-89% when compared with historical controls. Several other groups also reported similar improvements in outcomes, based on historical controls. A recent comparison using nonrandomized, but contemporary, controls by Tabbutt and colleagues revealed no difference in hospital survival.
The general trend in the literature has been that centers that have had difficulties in achieving optimal survivals with the Norwood procedure have found a benefit to using the RVPAC, whereas institutions that have had better success with the Norwood procedure have found no improvement. The optimal source of pulmonary blood flow remains to be determined. Each source has inherent advantages and disadvantages, and the long-term, or even intermediate-term, effects are unknown. The MBTS results in significant diastolic run-off and potential coronary steal. The RVPAC avoids this run-off but adds the undesirable need to place an incision on the right ventricle.
A multi-institutional, randomized, controlled trial comparing the MBTS and the RVPAC was recently concluded by the Pediatric Heart Network with funding from the National Heart, Lung, Blood Institute of the National Institutes of Health. The main results manuscript of the Single Ventricle Reconstruction trial are anticipated to be published in the summer of 2010.
Whichever source of pulmonary blood flow is selected, these procedures provide adequate pulmonary blood flow while decreasing volume overload to the right ventricle and improving effective pulmonary blood flow until the patient can undergo a completion Fontan procedure. As described in detail in Second-stage palliation: Hemi-Fontan or bidirectional Glenn anastomosis, the hemi-Fontan procedure is a modification of the bidirectional Glenn procedure. The hemi-Fontan procedure involves (1) a side-to-side connection between the superior vena cava (SVC)/right atrial junction and the pulmonary arteries, (2) routine augmentation of the branch pulmonary arteries, and (3) temporary patch closure between the pulmonary arteries and the right atrium.
As mentioned above, a newer alternative method of staged repair is the hybrid procedure. The hybrid procedure provides the essential elements of the Norwood procedure, namely, providing unrestricted systemic output from the right ventricle and controlling pulmonary blood flow without the need for an open heart procedure with cardiopulmonary bypass. For the hybrid procedure, the invasive cardiologist and the congenital cardiac surgeon work in concert. Generally, the surgeon opens the chest and places an introducer into the pulmonary artery. The cardiologist then places a stent in the patent ductus arteriosus to provide systemic output from the right ventricle. Concurrently or at interval, the atrial septal communication is assured by balloon septostomy +/- stenting and pulmonary artery blood flow is controlled with the use of bilateral pulmonary artery bands.
The second stage, the so-called comprehensive stage II procedure, involves an aortic reconstruction as in the Norwood procedure and a hemi-Fontan or bidirectional Glenn as described above.
Intraoperative Details
First-stage palliation: Norwood procedure
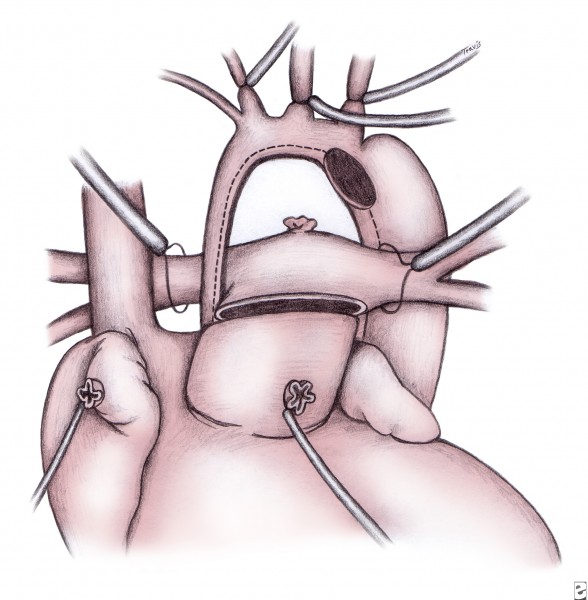
Through a midline sternotomy, cardiopulmonary bypass (CPB) is established. A minimum of 20 minutes of cooling to a core temperature of 18°C is begun for deep hypothermic circulatory arrest. Alternatively, some groups have reported the use of regional low-flow cerebral perfusion in lieu of deep hypothermic circulatory arrest (see Future and Controversies).
Regardless of the technique used, the septum primum is completely excised (atrial septectomy). The ductus is ligated and divided. The main pulmonary trunk is proximally divided to the bifurcation of the pulmonary arteries. The resultant opening in the pulmonary artery is closed with a patch of pericardium, polytetrafluoroethylene, or homograft. The remaining ductal tissue (on the undersurface of the aortic arch) is completely excised, and the incision is extended at least 10 mm further down the descending aorta into a normal-appearing and normal-caliber aorta (see following image).
This incision is proximally extended under the transverse arch and down the diminutive ascending aorta until the level of the previously divided main pulmonary trunk is reached (see following image).
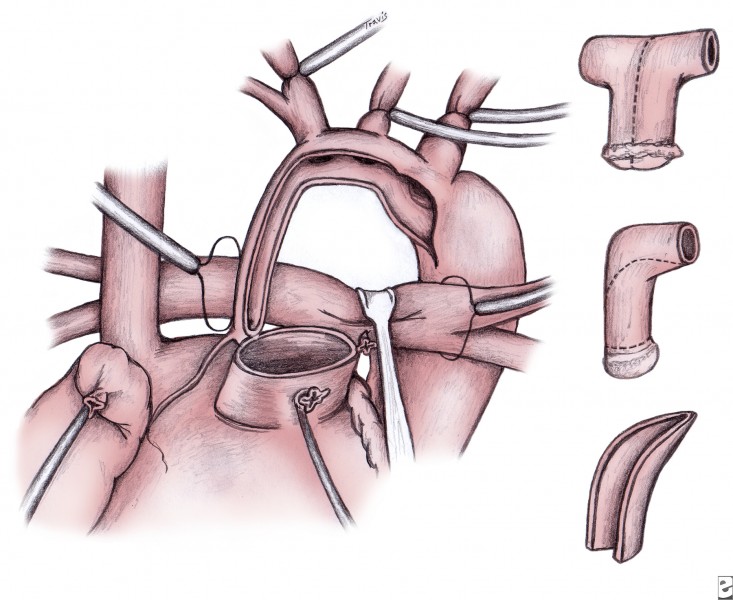
A cryopreserved pulmonary allograft is trimmed to fashion a patch that serves to enlarge the aorta and allow anastomosis to the proximal main pulmonary trunk (see inset in image above). The remainder of the aorta is attached to the pulmonary allograft, proximally incorporating the main pulmonary trunk. The cannulae are replaced to begin bypass and commence systemic rewarming to 37°C.
A polytetrafluoroethylene shunt is placed from the innominate artery to the central pulmonary artery during rewarming (see image below). A 4-mm shunt is used in patients who weigh more than 3.5-4 kg; smaller patients receive a 3.5-mm shunt. The distal end of the shunt is centrally placed on the pulmonary arteries, rather than onto the right pulmonary artery, to promote even distribution of blood flow to both lungs. Alternatively, the right ventricle–to–pulmonary artery conduit can be placed. These polytetrafluoroethylene grafts are generally either 5 mm or 6 mm in diameter.
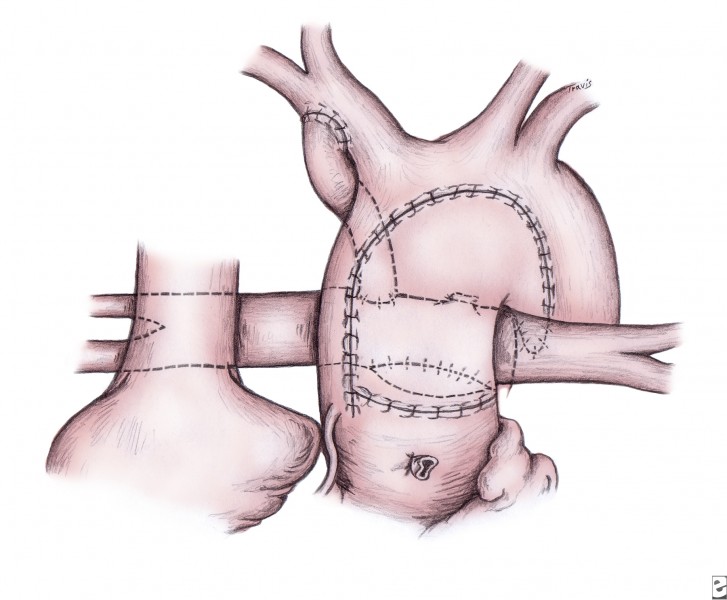 Completed Norwood procedure showing reconstructed neoaorta and modified Blalock-Taussig shunt from the innominate artery to the confluence of the branch pulmonary arteries. Image courtesy of Edward L. Bove, MD.
Completed Norwood procedure showing reconstructed neoaorta and modified Blalock-Taussig shunt from the innominate artery to the confluence of the branch pulmonary arteries. Image courtesy of Edward L. Bove, MD.
Second-stage palliation: Hemi-Fontan or bidirectional Glenn anastomosis procedure
The hemi-Fontan operation or a bidirectional Glenn anastomosis is typically performed in infants aged 3-10 months to minimize the period of time during which the right ventricle is subject to volume overload. Cardiac catheterization is performed prior to this procedure to evaluate pulmonary vascular resistance, pulmonary artery anatomy, tricuspid valve regurgitation, and right ventricular function.
To perform a bidirectional Glenn procedure, CPB is achieved with neoaortic arch cannulation and separate right-angle IVC and right-angle SVC cannulae. The aortopulmonary shunt is ligated and divided when CPB is initiated. If any stenosis of the pulmonary artery secondary to the prior shunt or patch is present, the stenosis is repaired with patch augmentation. The azygous vein is ligated and divided. The SVC is transected and anastomosed in an end-to-side fashion to the superior aspect of the right pulmonary artery. The cardiac end of the transected SVC is oversewn. Some groups routinely perform the bidirectional Glenn procedure without CPB, with or without an SVC-to–right aorta temporary shunt, during the anastomosis to minimize high cerebrovenous pressures.
The hemi-Fontan procedure has the same physiologic factors as a bidirectional Glenn anastomosis but includes an anastomosis of the pulmonary arteries to an incision in the atriocaval junction. The cavopulmonary connection may be performed under a brief period of deep hypothermic circulatory arrest. Alternatively, cannulation of the IVC and high on the SVC can be used to perform the procedure entirely during CPB.
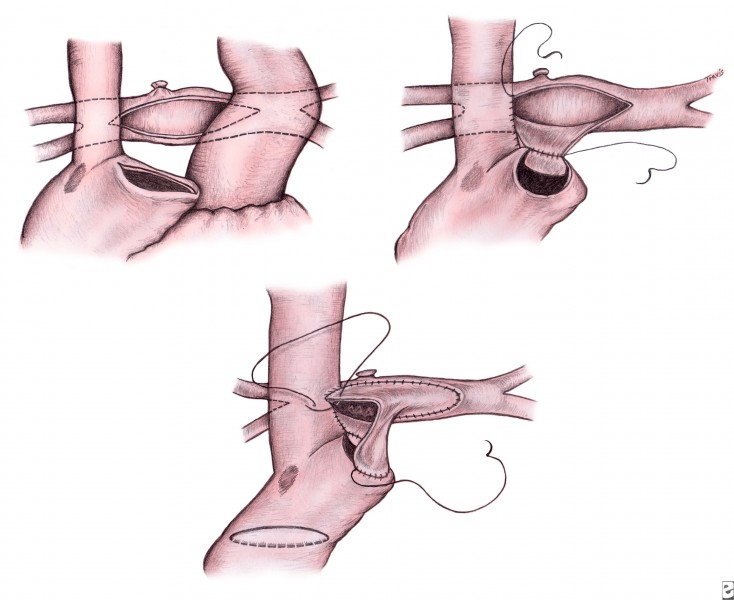
Whether the procedure is performed under circulatory arrest or during CPB, the remainder of the procedure is the same. The aortopulmonary shunt is divided, and the pulmonary arteries are mobilized from the right to the left upper lobe. The azygous vein is ligated. The right atrium is opened along the superior aspect of the appendage, and a corresponding incision is made transversely along the confluence of the branch pulmonary arteries (see upper left of image below). The posterior aspect of the right arteriotomy is anastomosed to the inferior aspect of the pulmonary arteriotomy (see upper right of following image).
A patch of pulmonary allograft tissue is fashioned to augment the pulmonary arteries. The allograft patch is begun at the left upper lobe, incorporating a separate end-to-side anastomosis for a left SVC, if necessary (see lower portion of above image). A patch is placed within the right atrium, which isolates SVC return into the pulmonary arteries and provides an unobstructed pathway for connection of IVC return during the Fontan procedure (see lower portion of above image). The atrial septal defect is inspected and enlarged, if necessary, which is completed best by cutting back the coronary sinus into the left atrium. Tricuspid valve repair is also performed as needed.
The advantage of the hemi-Fontan is that it shortens the length of time of CPB and dissection required for the completion Fontan procedure, which requires only the removal of the intra-atrial patch and placement of a lateral tunnel in the right atrium from the IVC to the SVC. In addition, routine augmentation of the branch pulmonary arteries helps optimize the anatomy for the completion Fontan procedure.
Third-stage palliation: Fontan procedure
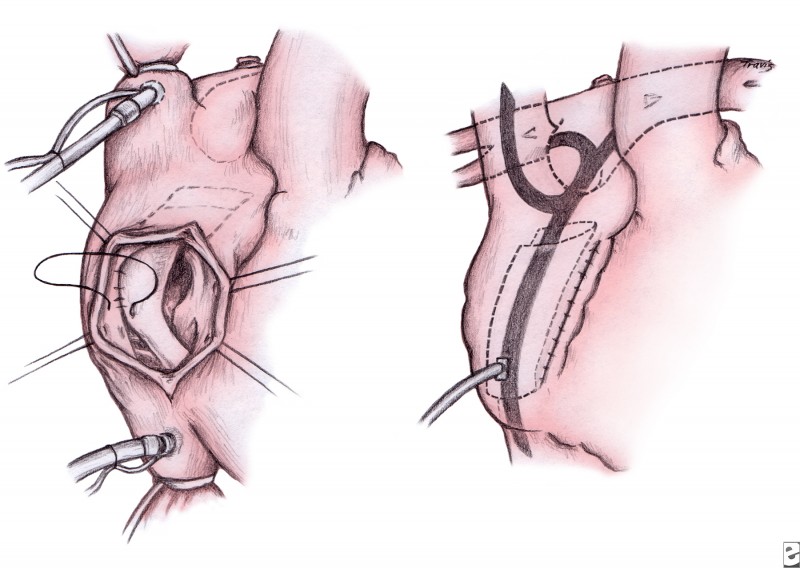
The completion Fontan procedure is usually performed in children aged 18-24 months. The infant is evaluated using cardiac catheterization prior to surgery. The Fontan technique used by the authors for HLHS anatomy is the technique termed total cavopulmonary connection with a lateral tunnel. After achieving CPB, the right atrium is opened.
If a hemi-Fontan procedure has been performed, the intra-atrial baffle is resected. In bidirectional Glenn anastomosis, a right atrium–to–pulmonary artery anastomosis is created. A baffle of polytetrafluoroethylene is fashioned and placed inside of the right atrium to convey the IVC return to the cavopulmonary connection (see image below). This technique minimizes the possibility of obstruction of the pulmonary venous return, which can be caused by an atriopulmonary anastomosis. Fenestration of the baffle may help prevent complications in high-risk patients and shorten the period of pleural drainage. Some centers opt for an extracardiac, instead of an intracardiac, lateral tunnel Fontan.
First-stage palliation: Hybrid procedure
The hybrid procedure is performed either in an operating room with cardiac catheterization capability or in a cardiac catheterization suite. A median sternotomy is performed and bilateral pulmonary artery bands are placed at the take off of the left pulmonary artery and on the right pulmonary artery between the aorta and superior vena cava. The bands are constructed by cutting a 1- to 2-mm ring from a 3.5-mm polytetrafluoroethylene tube graft (GoreTex, W. L. Gore & Associates, Inc, Flagstaff, Ariz). A 3.0-mm graft is used for patients weighing less than 2.5 kg.
The exact diameter of the bands is based upon patient weight, pulmonary artery diameter, systemic blood pressure, oxygen saturation, and angiography. Typically, systemic blood pressure increases by 10 mm Hg and peripheral oxygen saturation has an absolute decrease of 10% with appropriately tight bands. An introducer sheath is then placed through the main pulmonary artery via a pursestring suture. A stent is deployed in the patent ductus arteriosus to provide unrestricted systemic blood flow. Just prior to discharge, a balloon atrial septostomy, and, if necessary, a stent are performed to maintain the atrial communication.
Second-stage palliation: Comprehensive stage II procedure
Via a median sternotomy, cardiopulmonary bypass is initiated with cooling for deep hypothermic circulatory arrest or regional cerebral perfusion at the discretion of the surgeon. The pulmonary artery bands are removed and the arteries are augmented as needed. The stent is removed and the aortic reconstruction is performed as in a Norwood operation. A bidirectional Glenn or hemi-Fontan is performed, depending on surgeon preference.
Postoperative Details
First-stage palliation: Norwood procedure
After weaning from CPB, an atrial-monitoring catheter is placed to measure central venous pressure, and infusion of inotropes is initiated. University of Michigan medical staff routinely use continuous infusions of milrinone and low-dose dopamine, adding epinephrine in doses of 0.02-0.06 mcg/kg/min if hypotension is significant. Ventilation, with an initial FIO2 of 100% to achieve a PCO2 of approximately 35 mm Hg, is initiated and adjusted depending on the systemic arterial oxygen saturation and the systemic perfusion. If poor peripheral perfusion with systemic saturation in excess of 80-85% is noted, the FIO2 and minute ventilation are decreased to avoid excess pulmonary vasodilatation. The opposite maneuvers are used if systemic oxygen saturation is less than 70-75%.
Postoperative management is aimed at maintaining the delicate balance between the systemic and pulmonary vascular resistances and, therefore, relative systemic and pulmonary blood flow. Many regimens of ventilation, inotropic support, and vasodilatory support have been used, and multiple indicators of perfusion adequacy (mixed venous oxygen, lactate) have been measured with varying degrees of success.
Ideally, systemic arterial saturation should be maintained at 75-80%, which usually indicates that an optimal pulmonary-to-systemic blood flow ratio of less than 1 has been achieved. However, measurements of mixed venous oxygen saturation and pulmonary venous oxygen saturation are necessary to accurately assess the ratio of pulmonary blood flow (Qp) to systemic blood flow (Qs). The authors have found that serial lactate measurements provide an excellent indication of low cardiac output, and they rely on these determinations rather than mixed venous oxygen saturations.
Second- and third-stage palliation: Hemi-Fontan and Fontan procedures
For second- and third-stage operations, maintaining a low pulmonary vascular resistance is paramount. The pulmonary blood flow no longer is driven by an arterial shunt (or systemic RV, in cases of Sano modification), but by central venous pressure. Hypoxia and acidosis, which increase pulmonary vascular resistance, are avoided. Although mild respiratory alkalosis may be beneficial for pulmonary vascular resistance, a pH higher than 7.45-7.5 decreases cerebral blood flow and, hence, SVC return. After the hemi-Fontan procedure is performed, this decrease in SVC return decreases pulmonary blood flow. Inhaled nitrous oxide (NO) may also be used to decrease pulmonary vascular resistance but is infrequently needed.
Follow-up
After discharge from the hospital, regular cardiovascular evaluations are important. The child should be carefully observed for aortic arch obstruction, tricuspid insufficiency, and increasing cyanosis secondary to a limited atrial septal defect, shunt stenosis, or pulmonary artery distortion. For other long-term concerns, see Complications and Outcomes and Prognosis.
Complications
Complications that result from a procedure of the magnitude of the Norwood palliation are fairly common. Complications may include bleeding, low cardiac output syndrome, and arrhythmia in the immediate postoperative period. Aggressive correction of thrombocytopenia and coagulation factors is warranted. Poor peripheral and end-organ perfusion may represent poor cardiac output or pulmonary overcirculation, which may be treated by inotropic support or manipulation of relative pulmonary and systemic resistances. Common arrhythmias include junctional ectopic tachycardiac, which can be treated either with surface cooling to 35-36°C and pacing or with amiodarone infusion.
Incidence of unexplained sudden death, both in the immediate postoperative period and after discharge, remains problematic. In the postoperative period, the authors found that serial serum lactate determinations demonstrating failure to clear lactic acidosis have been helpful in predicting patients who will do poorly despite an apparently stable clinical condition.
Shunt complications, such as thrombosis, can occur. All patients are started on low-dose aspirin when they begin enteral nutrition. Other causes of increasing cyanosis during the postoperative period include pulmonary artery stenosis or distortion and restriction at the level of the atrial septal defect.
Evaluation prior to the hemi-Fontan procedure may reveal pulmonary artery stenosis, particularly of the left branch or at the insertion of the shunt. These stenoses are managed with patch augmentation during the hemi-Fontan procedure. Residual coarctation should be rare if the initial homograft patch is brought sufficiently onto the descending aorta during the Norwood procedure. Postoperative coarctation can usually be managed with balloon dilatation or, if necessary, surgical augmentation.
Long-term complications following the Fontan operation include atrial arrhythmia, thromboembolic events, and protein-losing enteropathy. Atrial arrhythmias are less common with the current techniques of cavopulmonary connections and may be treated with standard antiarrhythmic therapy. All of the authors’ patients who undergo the Fontan procedure are maintained on aspirin therapy, whereas others have advocated low-dose warfarin as prophylaxis against thromboembolism. Patients in whom the classic atriopulmonary connection Fontan procedure is performed with a dilated right atrium may benefit from conversion to a lateral tunnel or extracardiac Fontan operation to treat both arrhythmia and thrombosis.
Protein-losing enteropathy remains a difficult problem, affecting as many as 5% of patients who undergo the Fontan procedure. Treatment with intravenous infusions of albumin and immunoglobulin are supportive but not curative. Several other methods, including intravenous heparin, Fontan takedown, and cardiac transplantation, have been used with varying success.
Outcome and Prognosis
First-stage palliation
Bove et al at the University of Michigan studied first-stage palliation of hypoplastic left heart syndrome (HLHS) from January 1990 to August 1995 in 158 patients. All patients had classic HLHS, defined as right ventricle–dependent circulation, in association with atresia or severe hypoplasia of the aortic valve. Patients were subdivided into a standard-risk (n=127) population and a high-risk (n=31) population. High-risk patients included those undergoing the Norwood procedure after age 1 month, patients with severe obstruction to pulmonary venous return, and patients with significant noncardiac congenital conditions (ie, prematurity, low birth weight, chromosomal anomalies).
Hospital survivors numbered 120 (76%). The hospital survival rate was significantly better in the 127 standard-risk patients (86%) than in the high-risk group (42%). The risk factor analysis failed to reveal any effect on outcome by the morphologic subgroup, ascending aorta size, shunt size, initial pH at hospital presentation, or duration of circulatory arrest.
Among 151 patients at The Children’s Hospital of Philadelphia in a report by Norwood et al, 42 (28%) early deaths and 9 (5%) late deaths occurred. In a Children’s Hospital Boston series reported by Jonas et al, 78 neonates underwent palliative reconstructive surgery from 1983-1991. Hospital deaths numbered 29 (37%). Analysis of deaths revealed a greater risk of hospital death for infants with aortic atresia and mitral atresia, especially those with ascending aortic dimensions of less than 2 mm. However, in the authors’ experience, these conditions have not been associated with increased risk.
The results for the hospital survival for the Norwood procedure has continued to improve. In 2002, Tweddell and colleagues reported a 93% hospital survival in 81 patients undergoing a Norwood procedure for HLHS.
Galantowicz and colleagues published their results with the hybrid approach. The hospital survival rate was 97.5%.
Second-stage palliation
Hospital records of 114 patients undergoing the hemi-Fontan procedure for HLHS between August 1993 and April 1998 at the University of Michigan Medical Center were reviewed by Douglas et al. The overall hospital survival rate was 98% (112 patients). Sinus rhythm was present in 92% of patients. At the time of publication, 79 of the patients had undergone the completion Fontan procedure, with 74 survivors (94%). A similar study by Forbess et al from the Children’s Hospital Boston also revealed that a cavopulmonary anastomosis performed as a second-stage procedure for HLHS reduced mortality and improved intermediate survival rates.
Galantowicz and colleagues also reported their overall results on the 40 patients referenced above. There were 2 interstage deaths (5%), 2 reoperations, and 12 reinterventions in the catheterization lab. Thirty-six patients underwent comprehensive stage II procedures with 3 deaths (8%).
One hundred consecutive patients with classic HLHS underwent a Fontan procedure at the University of Michigan between February 1992 and April 1998. The survival rate in patients (n=52) undergoing surgery in the second half of the study and treated with a prior hemi-Fontan procedure at second-stage palliation was 98%. No deaths have occurred in patients undergoing the last 125 consecutive Fontan procedures for HLHS. Several other centers also have reported significant improvements in survival rates following the Fontan procedure in patients with HLHS.
The results of the hybrid approach through the Fontan procedure were also reported by Galantowicz et al. One interstage mortality occurred and 15 patients had undergone Fontan completion while 17 awaited Fontan. Among the 15 patients who underwent Fontan completion, no mortalities occurred.
Neurodevelopmental outcomes
As survivals have improved, other endpoints, such as patient neurodevelopmental outcome, have become of increasing interest to the healthcare provider caring for the patient with HLHS. Similar to any patient with cyanotic congenital heart disease, patients with HLHS are at risk for neurodevelopmental delay for multiple reasons. Cyanosis, congestive heart failure, and CNS abnormalities are associated with HLHS and can contribute to developmental delay. In addition, CPB and hypothermic circulatory arrest at the time of repair can cause neurologic injury.
In a recent study from the University of Michigan Medical Center, Goldberg and colleagues evaluated 51 patients with single ventricle physiology, 26 patients with HLHS, and 25 patients with other cardiac anomalies. The primary testing methods were the Wechsler Preschool and Primary Scales of Intelligence, revised for children aged 34-87 months, and the Wechsler Intelligence Scale, third edition, for children aged 72 months to 17 years. Additional tests included the Bayley Scales of Infant Development, the Vineland Adaptive Behavior Scales, and the Child Behavior Checklist.
Results indicated that children with HLHS scored statistically lower than children without HLHS with single ventricles. However, neither group scored significantly differently than population standards. As has been seen in children with congenital heart disease in general, patients in this study scored significantly better on tests of verbal intelligence than on tests of motor skills. Socioeconomic status, hypothermic circulatory arrest, and perioperative seizures were significant risk factors for impaired neurodevelopmental outcome. Duration of CPB, cardiac arrest requiring resuscitation, and clinical shock or pH less than 7.1 did not correlate with a poor neurodevelopmental result.
Future and Controversies
Several groups have begun to use the techniques of regional cerebral perfusion for aortic arch reconstruction in lieu of deep hypothermic circulatory arrest. For this technique, the proximal anastomosis of the modified Blalock-Taussig shunt is performed prior to arresting the heart. Then, the arterial cannula can be placed into the shunt, and perfusion is administered to the innominate artery. Whether these techniques will improve perioperative survival rates or long-term neurodevelopmental outcomes has yet to be determined. Recent publications have failed to demonstrate any improvement in outcome with regional cerebral perfusion.
Currently, numerous groups are advocating the use of an extracardiac conduit to complete the Fontan procedure. This technique may offer significant advantages; however, patients may be exposed to the risks of thromboembolic complications inherent in prosthetic conduits in the venous system. Lack of growth is also of concern. The literature does not have a consensus that favors one technique over the other.
The role of the hybrid approach compared to the traditional 3-stage method remains to be determined. Survivals at the most experienced centers are comparable. Whether there will be long-term benefit to avoiding the Norwood operation at the time of birth for such outcomes as neurodevelopment remain to be proven.
Comparison of the MBTS and the RVPAC awaits the results of the Single Ventricle Reconstruction trial, which should be published in the summer of 2010.
Future considerations for the Fontan procedure in this subgroup of patients include minimization of thromboembolic events, preservation of right ventricular and tricuspid valve function, and prevention of arrhythmias.