 Children born with cardiac and other congenital birth defects have potentially been linked to use of the SSRI class of antidepressant drugs, including Celexa (Generic: citalopram). Research suggests serious Celexa side effects, including the increased risk of congenital heart defects and other congenital birth defects. This link between Celexa (citalopram) and congenital heart defects has many health professionals concerned that pregnant women should not be prescribed the medication Celexa (citalopram) or other SSRIs. In addition to serious congenital Heart Birth Defects, SSRIs like Celexa (citalopram), Prozac, Zoloft and Paxil have also been linked to increased risk of Persistent Pulmonary Hypertension of the Newborn (PPHN).
Children born with cardiac and other congenital birth defects have potentially been linked to use of the SSRI class of antidepressant drugs, including Celexa (Generic: citalopram). Research suggests serious Celexa side effects, including the increased risk of congenital heart defects and other congenital birth defects. This link between Celexa (citalopram) and congenital heart defects has many health professionals concerned that pregnant women should not be prescribed the medication Celexa (citalopram) or other SSRIs. In addition to serious congenital Heart Birth Defects, SSRIs like Celexa (citalopram), Prozac, Zoloft and Paxil have also been linked to increased risk of Persistent Pulmonary Hypertension of the Newborn (PPHN).
Antidepressant Drugs Include:
What is Celexa?
Celexa (Generic: citalopram) is an antidepressant drug of the selective serotonin reuptake inhibitor (SSRI) class. It has FDA approval to treat major depression, and is prescribed off-label for a number of anxiety conditions. Citalopram (pronounced /saɪˈtælɵpræm/) was originally created in 1989 by the pharmaceutical company Lundbeck. The patent expired in 2003, allowing other companies to legally produce generic versions. Lundbeck has recently released an updated formulation called escitalopram (also known as Cipralex or Lexapro), which is the S-enantiomer of the racemic citalopram, and acquired a new patent for it. In the United States, Forest Labs manufactures and markets the drug.
Celexa (citalopram) is approved to treat the symptoms of major depression. Celexa (citalopram) is frequently used off-label to treat anxiety, panic disorder, ADHD, PMDD, Body dysmorphic disorder and OCD. Celexa (citalopram) has been found to reduce the symptoms of diabetic neuropathy. Celexa (Citalopram) and other SSRIs like Lexapro, Prozac, Paxil and Zoloft can be used to treat hot flashes.
Citalopram is sold under the brand-names Celexa (U.S. and Canada, Forest Laboratories, Inc.), and Citalopram (USA, United Kingdom, Denmark). Celexa (Citalopram) is available in 10 mg, 20 mg, and 40 mg tablets, as well as 10 mg/5 mL peppermint flavor oral solution.
Celexa Birth Defects
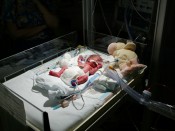 Celexa (citalopram) has been linked to increased risk of serious birth defects including Primary Pulmonary Hypertension of the Newborn (PPHN). A study published in the New England Journal of Medicine revealed that newborns whose mothers took SSRI antidepressants such as Celexa during pregnancy were six times more likely to be born with the cario-pulmonary birth defect PPHN than those whose mothers did not take prescribed antidepressants – PPHN rose as high as 12 cases per 1,000 births.
Celexa (citalopram) has been linked to increased risk of serious birth defects including Primary Pulmonary Hypertension of the Newborn (PPHN). A study published in the New England Journal of Medicine revealed that newborns whose mothers took SSRI antidepressants such as Celexa during pregnancy were six times more likely to be born with the cario-pulmonary birth defect PPHN than those whose mothers did not take prescribed antidepressants – PPHN rose as high as 12 cases per 1,000 births.
Another study published in Pediatrics (February, 2004) found that pregnant women who used SSRIs had an increased risk of giving birth to babies with abnormal heart rhythms, unusual sleeping patterns, disrupted neurological development and problems with alertness.
A study summarized on Web MD reported that newborns exposed to SSRI antidepressants such as Celexa by their pregnant mothers late in pregnancy were twice as likely to risk admission to special-care nurseries and twice as likely to suffer respiratory complications serious enough to require hospital ventilation procedures.
In addition to PPHN, SSRI antidepressants like Celexa, Paxil, Prozac and Zoloft have been linked to severe congenital heart defects, including: atrial septal defects (ASD – hole in the heart), ventricular septal defects (VSD – hole in the heart), tetrology of fallot (ToF), hypoplastic left heart syndrome (HLHS), transposition of the great arteries (TGA or TOGA), patent ductus arteriosus (PDA), total anomalous pulmonary venous return (TAPVR), coarctation of the aorta (CoA), double outlet right ventricle (DORV), and Shone’s Complex. These injuries range from minor heart murmurs not requiring surgery, all the way to the most severe congenital heart defects, requiring multiple surgeries or potentially a complete heart transplant in order to save the life of the child..
Celexa Side Effects
 Celexa (citalopram) theoretically causes side effects by increasing the concentration of serotonin in other parts of the body (e.g., the intestines). Common side effects of citalopram include drowsiness, insomnia, nausea, weight changes, frequent urination, decreased sex drive, anorgasmia, dry mouth, increased sweating, trembling, diarrhea, excessive yawning, and fatigue. Less common side effects include bruxism, vomiting, cardiac arrhythmia, blood pressure changes, dilated pupils, anxiety, mood swings, headache, and dizziness. Rare side effects include convulsions, hallucinations, and severe allergic reactions. Celexa (Citalopram) is also a mild antihistamine, which may be responsible for some of its sedating properties. If sedation occurs, the dose may be taken at bedtime rather than in the morning. Other side effects, such as increased apathy and emotional flattening, may be caused by the decrease in dopamine release that is associated with increased serotonin.
Celexa (citalopram) theoretically causes side effects by increasing the concentration of serotonin in other parts of the body (e.g., the intestines). Common side effects of citalopram include drowsiness, insomnia, nausea, weight changes, frequent urination, decreased sex drive, anorgasmia, dry mouth, increased sweating, trembling, diarrhea, excessive yawning, and fatigue. Less common side effects include bruxism, vomiting, cardiac arrhythmia, blood pressure changes, dilated pupils, anxiety, mood swings, headache, and dizziness. Rare side effects include convulsions, hallucinations, and severe allergic reactions. Celexa (Citalopram) is also a mild antihistamine, which may be responsible for some of its sedating properties. If sedation occurs, the dose may be taken at bedtime rather than in the morning. Other side effects, such as increased apathy and emotional flattening, may be caused by the decrease in dopamine release that is associated with increased serotonin.
Celexa Class Action Lawsuit vs. Individual Celexa Lawsuit
There are distinct differences between a Celexa (citalopram) class action lawsuit and a more typical individual Celexa lawsuit. A Celexa class action lawsuit would be a form of Celexa lawsuit in which a large group of people (plaintiffs) collectively bring a lawsuit to court in the form of a “class action” against the manufacturers of Celexa (defendant). In a class action lawsuit involving personal injury, resulting from defective products such as antidepressant SSRI drugs like Paxil (paroxetine), Zoloft (sertraline), Prozac (fluoxetine) and Celexa (citalopram), all Celexa lawsuit plaintiffs would typically be grouped together into a single Celexa class action lawsuit, regardless of the degree or severity of their birth defect injuries. In this type of Celexa class action lawsuit, plaintiffs with injuries ranging from minor heart murmurs not requiring surgery, all the way to the most severe congenital heart defects, requiring multiple surgeries or a complete heart transplant, would be grouped into one single Celexa class action lawsuit. All plaintiffs in the class would equally share any award or settlement resulting from the Celexa class action lawsuit.
In Celexa lawsuits involving catastrophic injury or death, an individual lawsuit, in most cases, is more appropriate and in the plaintiff’s best interest. SSRI antidepressants like Celexa, Paxil, Prozac and Zoloft have been linked to severe congenital heart defects, including: atrial septal defects (ASD – hole in the heart), ventricular septal defects (VSD – hole in the heart), tetrology of fallot (ToF), hypoplastic left heart syndrome (HLHS), transposition of the great arteries (TGA or TOGA), patent ductus arteriosus (PDA), total anomalous pulmonary venous return (TAPVR), double outlet right ventricle (DORV), and coarctation of the aorta (CoA). Celexa cases such as these are better suited to an individual Celexa lawsuit because of the severity and degree of injury to the plaintiff. In an individual Celexa Lawsuit, each plaintiff’s case is filed, presented and considered individually, based on its own strength and degree of injury.
In many cases involving SSRI antidepressants like Celexa, Paxil, Prozac and Zoloft and the serious congenital heart defects related to these SSRI antidepressants, surgery is required. Heart surgery will typically be required when a child is an infant or toddler and then again, potentially multiple times, as the child grows to maturity. In many cases, with surgery and medical care, children may be able to lead mostly normal and productive lives. An individual Celexa lawsuit allows each Celexa victim, their injuries and their future needs to be considered on an individual basis when determining damages, awards and settlement amounts, and not as part of a class action lawsuit.
Celexa Autism
A recent study published by Archives of General Psychiatry has found a link between antidepressant use during pregnancy and Autism Spectrum Disorder (ASD). Pregnant women taking Celexa antidepressants during the first trimester of pregnancy may triple the chances of their child having Celexa autism. This study was prompted when researchers noticed a rise over the past few years in the use of selective serotonin reuptake inhibitor antidepressant (SSRIs) and an increase in Celexa autism diagnoses.
Celexa Autism Side Effects
- Autism from Celexa
- ASD – Autism Spectrum Disorder
- PDD – Pervasive Developmental Disorder
- PDD – NOS – Pervasive Developmental Disorder – Not Otherwise Specified
If you or a loved one took Celexa while pregnant and have a child that may suffer Celexa autism or other Celexa birth defects, contact our law firm immediately to discuss the legal options available to you and your family.
See also: Autism Lawsuit: Antidepressants During Pregnancy Increase Autism Risk
Speak to a Celexa Lawyer
If you took Celexa or generic citalopram during pregnancy and your child was born with a birth defect, we encourage you to contact a Celexa litigation attorney at our law firm immediately. It may be too late to recover from the debilitating effects of Celexa, but an experienced products liability attorney at the Willis Law Firm can assist you in a legal action against the makers of Celexa. You are not alone. Join other birth defect victims and their families in speaking up and fighting for your legal rights.
Celexa Lawsuit
Please fill out our free online legal evaluation form and we will contact you within 24 hours. Please keep in mind that certain states have statutes of limitation that limit the amount of time you have to file a lawsuit or seek legal action. Contact our law firm immediately so that we may explain the rights and options available to you and your family.

