; ?>/images/no-longer-accepting-cases.jpg” alt=”Previously Investigated Drugs – No Longer Accepting Cases” /></div>
<p>At this time our law firm is no longer accepting new cases involving this drug. This does not mean that you do not have a case; please seek a second opinion with local legal counsel to discuss your options.</p>
<hr/>
<h2>What is Remeron?</span></h2>
<div><a rel=)
 Remeron (mirtazapine) is a tetracyclic antidepressant. It affects chemicals in the brain that may become unbalanced and cause depression. It is thought to increase the activity of norepinephrine and serotonin which help elevate mood. Remeron is used to treat major depressive disorder. Remeron comes as a tablet and as a disintegrating tablet to taken by mouth. It usually is taken once a day at bedtime. Remeron is sometimes prescribed for other uses.
Remeron (mirtazapine) is a tetracyclic antidepressant. It affects chemicals in the brain that may become unbalanced and cause depression. It is thought to increase the activity of norepinephrine and serotonin which help elevate mood. Remeron is used to treat major depressive disorder. Remeron comes as a tablet and as a disintegrating tablet to taken by mouth. It usually is taken once a day at bedtime. Remeron is sometimes prescribed for other uses.Remeron Side Effects
- Agitation, hallucinations, overactive reflexes, nausea, vomiting, diarrhea
- Chest pain, tachycardia
- Seizures
- Loss of coordination, fainting
- White patches or sores inside your mouth or on lips
- Trouble concentrating, memory problems, weakness, unsteadiness, confusion
- Fever, chills, sore throat, mouth sores, and other signs of infection
- Risk of suicide
- Birth defects
Remeron Birth Defects
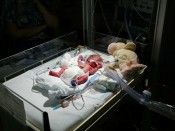 The FDA has classified Remeron (mirtazapine) as a pregnancy category “C” drug, pending further birth defect investigation and studies. At the present time, animal birth defect studies have yet to conclusively document the teratogenic effects of Remeron. With regard to birth defects, medical experts stress that Remeron (mirtazapine) should only be given during pregnancy when benefits outweigh risks for potential birth defects. Various classes of antidepressants have long been linked to serious congenital birth defects. SSRI antidepressants have been linked to severe congenital heart defects and lung defects, including: atrial septal defects (ASD – hole in the heart), ventricular septal defects (VSD – hole in the heart), tetrology of fallot (ToF), hypoplastic left heart syndrome (HLHS), transposition of the great arteries (TGA or TOGA), patent ductus arteriosus (PDA), total anomalous pulmonary venous return (TAPVR), coarctation of the aorta (CoA), and persistent pulmonary hypertension of the newborn (PPHN).
The FDA has classified Remeron (mirtazapine) as a pregnancy category “C” drug, pending further birth defect investigation and studies. At the present time, animal birth defect studies have yet to conclusively document the teratogenic effects of Remeron. With regard to birth defects, medical experts stress that Remeron (mirtazapine) should only be given during pregnancy when benefits outweigh risks for potential birth defects. Various classes of antidepressants have long been linked to serious congenital birth defects. SSRI antidepressants have been linked to severe congenital heart defects and lung defects, including: atrial septal defects (ASD – hole in the heart), ventricular septal defects (VSD – hole in the heart), tetrology of fallot (ToF), hypoplastic left heart syndrome (HLHS), transposition of the great arteries (TGA or TOGA), patent ductus arteriosus (PDA), total anomalous pulmonary venous return (TAPVR), coarctation of the aorta (CoA), and persistent pulmonary hypertension of the newborn (PPHN).
The results of a prospective, comparative, observational study (n=312) indicates that 95% of the pregnant women were exposed to Remeron (mirtazapine) during the organogenesis period of pregnancy and 25% were exposed throughout the entire pregnancy. Among other potential effects, exposure to mirtazapine was also associated with a higher rate of preterm births.
Remeron Class Action Lawsuit vs. Individual Remeron Lawsuit
There are distinct differences between a Remeron (mirtazapine) class action lawsuit and a more typical individual Remeron lawsuit. A Remeron class action lawsuit would be a form of Remeron lawsuit in which a large group of people (plaintiffs) collectively bring a lawsuit to court in the form of a “class action” against the manufacturers of Remeron (defendant). In a class action lawsuit involving personal injury, resulting from defective products such as antidepressant SSRI drugs like Remeron (mirtazapine), Paxil (paroxetine), Prozac (fluoxetine) and Celexa (citalopram), all Remeron lawsuit plaintiffs would typically be grouped together into a single Remeron class action lawsuit, regardless of the degree or severity of their birth defect injuries. In this type of Remeron class action lawsuit, plaintiffs with injuries ranging from minor birth defects not requiring surgery, all the way to the most severe congenital heart defects, requiring multiple surgeries or potentially a complete heart transplant, would be grouped into one single Zoloft class action lawsuit. All plaintiffs in the class would equally share any award or settlement resulting from the Remeron class action lawsuit.
In Remeron lawsuits involving catastrophic injury or death, an individual lawsuit, in most cases, is more appropriate and in the plaintiff’s best interest. Antidepressants like Remeron, Zoloft, Prozac, Celexa and Paxil, have been linked to severe congenital heart defects, including: atrial septal defects (ASD – hole in the heart), ventricular septal defects (VSD – hole in the heart), tetrology of fallot (ToF), hypoplastic left heart syndrome (HLHS), transposition of the great arteries (TGA or TOGA), patent ductus arteriosus (PDA), total anomalous pulmonary venous return (TAPVR), and coarctation of the aorta (CoA). Potential Remeron cases such as these are better suited to an individual Remeron lawsuit because of the severity and degree of injury to the plaintiff. In an individual Remeron Lawsuit, each plaintiff’s case is filed, presented and considered individually, based on its own strength and degree of injury.
In many cases involving antidepressants like Remeron, Zoloft, Prozac, Celexa, Paxil and the serious congenital birth defects related to these antidepressants, surgery is required. Surgery may be required when a child is an infant or toddler and then again, potentially multiple times, as the child grows to maturity. In many cases, with surgery and medical care, children may be able to lead mostly normal and productive lives. An individual Remeron lawsuit allows each Remeron victim, their injuries and their future needs to be considered on an individual basis when determining damages, awards and settlement amounts, and not as part of a Remeron class action lawsuit.
Remeron Birth Defect Lawsuit
If you took Remeron or generic Mirtazapine during pregnancy and your child was born with a severe birth defect, we encourage you to contact a Remeron litigation attorney at our law firm immediately. It may be too late to recover from the devastating effects of Remeron, but an experienced products liability attorney at the Willis Law Firm can assist you in legal action against the makers of Remeron. You are not alone. Join other birth defect victims and their families in speaking up and fighting for your legal rights.
Please fill out our free online legal evaluation form and we will contact you within 24 hours. Please keep in mind that certain states have statutes of limitation that limit the amount of time you have to file a lawsuit or seek legal action. Contact our law firm immediately so we may explain the rights and options available to you and your family.

